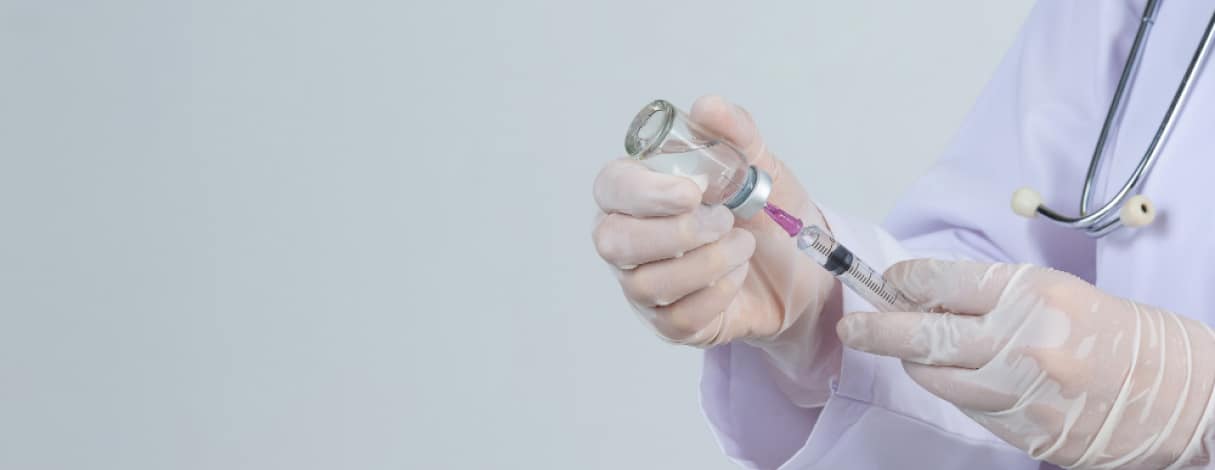
In aesthetic medicine, achieving the desired look often requires a strategic approach, including the need to reverse previous treatments. Enter Liporase, an enzyme-based filler dissolver that has become popular for its ability to safely and effectively correct unwanted filler results. This article will focus on the proper use of Liporase, ensuring safe and effective outcomes for both practitioners and patients.
Liporase, containing the enzyme hyaluronidase, is a valuable tool in aesthetic medicine for reversing hyaluronic acid-based fillers. It mitigates fibrosis and necrosis risks and stimulates fibroblast activity. Known for its effectiveness, versatility, and safety, Liporase should only be used by trained and certified professionals.
Common side effects include redness, swelling, bruising, and discomfort. Severe complications, though less frequent, can include infection, allergic reactions, and tissue damage.
Results are often immediate, but full effects can take up to 48 hours. Monitor patients for adverse reactions and provide aftercare instructions, such as:
Liporase is a powerful tool for safely and effectively dissolving dermal fillers, but it should only be administered by certified professionals. Proper training, ethical considerations, and effective patient communication are key to successful outcomes.
Join our newsletter to receive latest news and offers

Medicle MD Ltd
Reg. Number: 14317237
Address: 27 Old Gloucester Street,
WC1N 3AX London,
United Kingdom
Our website is intended solely for people who use medical devices, such as dermal fillers, as professionals. It may contain product advertisements targeted only at such people. To enter Nu Derma Supply, please confirm that you are such a person (e.g. a medical professional, cosmetologist, service technician, etc.).